A Brief History
In 1869 Dmitri Mendeleev, a Russian chemist, designed a way of organizing the elements known at the time. He set them up in the order of atomic weight, and further organized them into rows and columns based on their physical and chemical properties. At this time, Mendeleev had little to no knowledge on the properties of atoms. Nonetheless, he still managed to compose the periodic table extremely similar to the one we know today. But in 1869, some elements on our current periodic table were unknown, so they were not in his original table.
Since some elements were unknown, there were gaps in the periodic table. Based on Mendeleev's intelligence, he was able to determine that there were, in fact, many new elements that existed and the properties of these unknown.
Mendeleev had a main rule to categorize the elements:
Elements in any of the groups of columns of the periodic table are similar to the other elements in their column. Elements are in the location that they are because of the way they act and look. The periodic table is like a grid, therefore there are columns (up and down) and rows (left to right). The rows are considered to be different periods and columns are considered to be different groups:
Elements in any of the groups of columns of the periodic table are similar to the other elements in their column. Elements are in the location that they are because of the way they act and look. The periodic table is like a grid, therefore there are columns (up and down) and rows (left to right). The rows are considered to be different periods and columns are considered to be different groups:
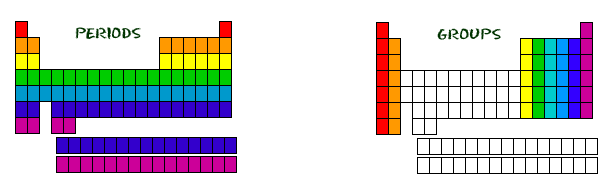
When considering the period, all of the rows go left to right. The first row has it's own period, the second row has its own, and as you go down the rows, they each have their own period. In the table, if two elements are considered to be similar, they are located in the same row, and therefore have the same period. The period signifies the quantity of atomic orbitals the element has. In the picture to the left, the elements in red (the first period) has one orbital for its electrons. The orange elements have two orbitals, the yellow have three, and the number gets consecutively larger as you go down the rows. Seven is the maximum number of electron orbitals, and there are only seven rows.
Columns are very similar to rows, and the different columns signify different groups in the table. If two elements are in the same group, that means that they have the same amount of electrons in their most outer orbital, most commonly known as valence electrons. In the picture to the right, all of the red elements have one valence electron, all of the orange have two, and the number gets consecutively higher as you move left to right. But, this rule only works if you exclude the transition metals (the elements not colored in). These elements start to add electrons to their second-to-last shell instead. This is possible because, transition metals are able to put more than eight electrons in the second-to-last shell, they are able to put up to 32, but none can have more than 32.