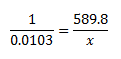What are the units for ionization energy?
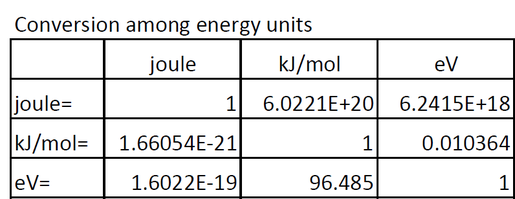
The most commonly used units for ionization energy are:
1. eV [electron volts] (in Physics)
2. kJ/mol [molar units] (in Chemistry)
1. eV [electron volts] (in Physics)
2. kJ/mol [molar units] (in Chemistry)
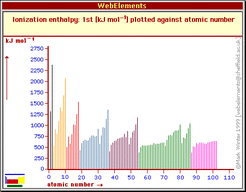
- kJ/mol is the the amount of energy needed to remove an electron from every atom in a particular mole of atoms. There is a specific amount of energy needed, because there is an attractive force that pulls the electron to the nucleus; thus this has to be removed. Measuring this way is called molar ionization energy or enthalpy.
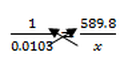
- An electron volt is the amount of energy produced by 1 mole of charge, which is (6.022x10^23), when you change the electrical potential difference (electrical potential energy) of 1 electron one volt 96.485 kJ/mol.