What is the pattern?
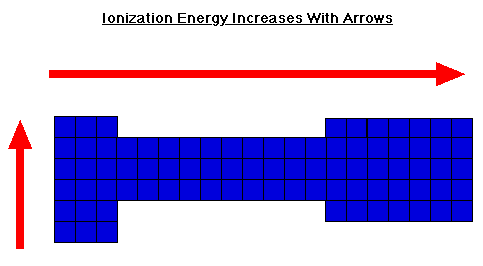
We know that ionization energies involve ions, atoms which have lost or gained electrons. We have already looked at the periodic table in relation to it; so, what is the trend for IE?
Ionization Energy values increase as you go from bottom to top and as you go from left to right.
Ionization Energy values increase as you go from bottom to top and as you go from left to right.
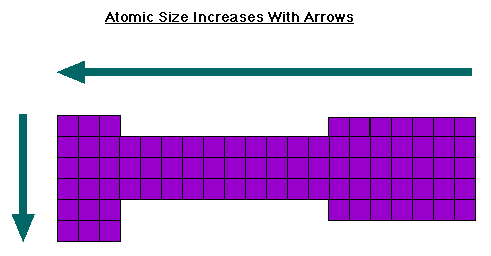
Why does this trend exist?
Why does this trend exist? Well something that also relates to the ionization energy trends is the atomic size trend. Oppositely to IE, atomic size increases from right to left and top to bottom. This makes sense because as the atoms get smaller, the valence electrons get closer to the nucleus, therefore the harder it becomes to remove (because IE is the energy required to completely remove an electron from a gaseous atom or ion).